Ex-OR Tambo Municipality accountant guilty in R9.8m fraud case
A former OR Tambo District Municipality capital expenditure accountant will be sentenced in March after she was this week convicted of fraud and money laundering charges.
The Umtata High Court on Wednesday convicted Nyameka Qongqo of the charges emanating from a 2014 case when the accused was still an employee at OR Tambo Municipality.
In a statement, the Hawks said Qongqo effected duplicate payments to the value of R9.8 million to her husband’s business account, Zingce Trading, as well as her sister’s business account, Ogiyonke Construction.
“The case was reported to the Hawks for investigation and upon conclusion of the investigation, five people, including juristic persons, were charged with fraud and money laundering. Qongqo was yesterday found guilty on both charges,” the Hawks said.
Qongqo’s sentencing was postponed to 25 March 2021, while she is out on bail.
Meanwhile, Qongqo’s husband, Thobani Alex Ntshokovu, and his company were convicted early in 2020 and sentenced to five years wholly suspended and to further pay an amount of R50 000 as a fine.
The case is still continuing against the accused’s sister and her company, and is also remanded to 25 March 2021.
The Serious Corruption Crime Investigation unit was applauded by the Eastern Cape Provincial Head of the Directorate for Priority Crime Investigation, Mboiki Obed Ngwenya, for a job well done.
Application deadline for ECD relief fund extended
The Department of Social Development has extended the deadline for applications of the Early Childhood Development – Employment Stimulus Relief Fund (ECD-ESRF) to 26 February 2021.
In addition, a new web-form (available in three other languages over and above English) to assist with the applications is available on the www.ecd.gov.za website.
It will allow applicants to save and update their applications, as well as check their application status using their reference number.
The fund was established to assist ECD services recover from the loss of income brought on by COVID-19.
All types of ECD services (registered, conditionally registered or unregistered), including centre and non-centre based services, can apply for this relief funding.
Non-centre based services include play groups, toy libraries, child minders and mobile ECD programmes.
Furthermore, organisations that run, manage or oversee multiple centre and non-centre based ECD services may apply for more than one ECD programme and more than one type of ECD programme.
“As at 17 February 2021, there were a total of 16 114 applications received, with 74 525 ECD employees registered.
“As a further support mechanism, the department, in partnership with the DG-Murray Trust, has made available 135 non-governmental organisations and 2 236 Harambee youth (who are distributed throughout the country) to assist ECD services to apply,” the department said on Thursday.
The provincial Departments of Social Development, through their local offices, are also available to assist organisations.
Any ECD service that requires such assistance can visit their local Social Development office.
They can further send an email to ecdstimulus@dgmt.co.za or contact the call centre on 0800 089 666 and support will be provided.
“The department is encouraged by the response in the number of applications received to date and wishes to express its appreciation to the ECD sector and its partners in heeding the solidarity call to assist ECD services to access this much needed support.
“ECD services that have not yet applied are requested to do so by the extended closing date of 26 February 2021,” the department said.
Police seize unregistered medicines at OR Tambo International Airport
The South African Police Service (SAPS) this week seized unregistered medicines worth a market value of R126 million at the OR Tambo International Airport.
The unregistered medicines which are believed to be Ivermectin were mainly found to be in tablet form.
The discovery of the tablets was made by authorities during an inspection of a cargo container that was destined to South Africa from India.
Upon a compliance verification of the unregistered medicines by the South African Health Products Regulatory Authority (SAHPRA), authorities found that the company that had imported the medicines into the country, had done so without the necessary written approval and authorization from the Regulatory Authority.
While no arrests have been made as yet, the SAPS through its Interpol office are working with authorities from India to establish the exact delivery address of the consignment in the country.
The latest confiscation of the unregistered medicines is the largest bust concerning the illegal importation of medicines since the beginning of the year.
In January 2021, a multidisciplinary team arrested a total of seven people for the illegal importation of unregistered medicines mostly found to be Ivermectin.
One person was arrested at the King Shaka International Airport while six others were arrested at OR Tambo International Airport.
All seven accused have already appeared before court and are currently out on bail.
In all the cases, preliminary investigations revealed that the unregistered medicines had been imported for sale purposes and would have been utilized in the treatment of the COVID-19 virus.
“The SAPS therefore draws the attention of pharmaceutical companies, including health experts to section 22(c) of the Medicines Act which clearly states that, anyone who wishes to import medicines into the country must have written authority from SAHPRA.
“It should be further noted that those who do not comply with the Act will be apprehended and brought before a court of law to answer to illegal dealing and/or importation of unregistered medicines,” SAPS said in a statement.
Everything you need to know about the Johnson & Johnson vaccine
South Africa kick-started its first phase of the vaccination rollout programme using the Johnson & Johnson vaccine on Wednesday.
Yesterday, the first group of healthcare workers lined-up to receive the shot of the 80 0000 Johnson & Johnson vaccines that landed on Tuesday, which the department has described as a success.
The Health Department answers some of the burning questions about who will get it first; why it is a good choice for the country; why pregnant woman cannot get the shot and how effective it is?
Why is the Johnson & Johnson vaccine a good first choice for the National Vaccination Programme?
It was tested in a large trial of almost 44 000 people from four continents, of whom 7 000 participants came from South Africa.
The study also provided a good picture of how the vaccine works against the new 501Y.V2 variant, which is dominant in South Africa and currently responsible for around nine in 10 of all COVID-19 infections detected during the second wave.
The South African trial showed that while the Johnson & Johnson vaccine is not going to prevent mild symptoms, it provides 57% protection against moderate-severe disease, 85% protection against severe disease and 100% protection against death.
By way of comparison, the Oxford-AstraZeneca vaccine provided only 27% protection against mild to moderate COVID-19 caused by the new 501Y.V2 variant.
How does the Johnson & Johnson vaccine compare with other COVID-19 vaccines?
Like all COVID-19 vaccines, the jab contains instructions for the spike protein on the Coronavirus.
These instructions are delivered to the immune system by a modified adenovirus that has also been used in other vaccines, such as the Ebola and Zika shots.
The adenovirus is safe to use in vaccines as it has been modified so that it cannot cause disease or replicate in humans.
Non-active ingredients in the vaccine include sodium chloride, citric acid monohydrate buffer, polysorbate 80, 2 hydroxypropyl-β-cyclodextrin (HBCD), ethanol (absolute), sodium hydroxide and water for injection.
All of these ingredients are safe for human consumption.
The vaccine does not contain any animal products and is halal.
How does the vaccine protect you against COVID-19?
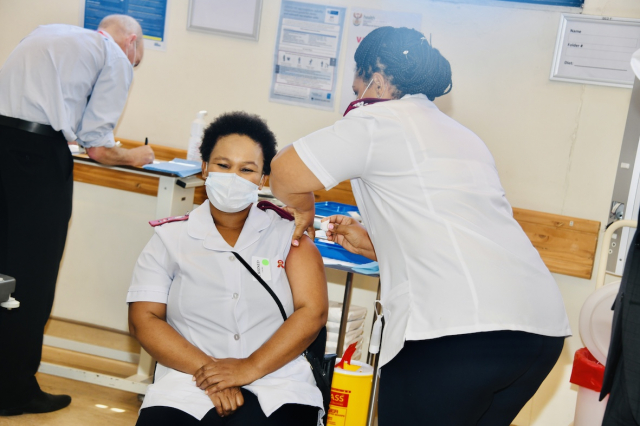
When injected, the vaccine trains your immune system to fight the virus that causes COVID-19.
The vaccine does not contain the Coronavirus and will not give you COVID-19.
Some mild side effects include tenderness at the injection site, feeling unwell, feverish and a headache for a few days and these are positive signs that the body is mounting an immune response to the Coronavirus.
When you encounter real Coronavirus particles in future, your immune system will be able to disarm the virus so that you either do not fall ill at all or if you do, the symptoms are mild and your chances of being hospitalised or dying are much lower.
How is it stored?
The vaccine has a long shelf life of two years at minus 20 degrees Celsius and can be safely stored in a domestic fridge for a month.
This makes it suitable for widespread rollout in warmer countries like South Africa as ultra-cold freezers are not needed.
How is the vaccine given and when does protection start?
The vaccine is given as a single dose into the upper arm.
Protection starts around 10 to 14 days after vaccination and even as early as seven days for severe disease and can also rise to good levels around a month after vaccination.
It is currently the only vaccine available that uses a single dose, while others require two shots.
No vaccine provides 100% protection. However, the jab is said to provide 57% protection against moderate-severe disease, 85% protection against severe disease, and 100% protection against death.
The researchers are still waiting for further results on how effective the vaccine is at preventing asymptomatic transmission.
It is important you continue to use masks, practise social distancing and good hand hygiene.
If you are a healthcare worker, you still need to wear your personal protective equipment (PPE) and stay vigilant, even after you are vaccinated.
Why is a research programme being used to make the vaccine available?
Manufacturers apply for vaccines to be licensed after trials show to be safe and effective.
The Johnson & Johnson trial results were released in early February and applications for licensing are underway in the United States, Europe and South Africa.
The Sisonke ‘Together’ programme allows the government to make this safe and effective vaccine immediately available to healthcare workers using a research study.
Sisonke is not the same as a clinical trial, rather a way that research can help to make it available while the licensing process takes place.
The South African government has chosen to move ahead with this programme because it would be unethical to withhold a vaccine known to be safe and effective.
The South African Health Products Regulatory Authority (SAHPRA) has approved the use of the vaccine for the programme while it processes the full licensing.
It is important to understand that the fact that it is not yet licenced does not mean that it is not safe or effective.
The third wave of COVID-19 is predicted to begin in South Africa this winter and protecting healthcare workers is a priority and so we must start vaccinating healthcare workers before the third wave arrives.
How is the Sisonke rollout going to work?
South Africa has secured 500 000 doses of the vaccine from Johnson & Johnson, enough to vaccinate half a million health workers.
The first batch of 80 000 doses arrived on Tuesday, 16 February and further deliveries will follow every two weeks.
Vaccines will be available at 17 hospitals throughout the country where teams of researchers and vaccinators will work together to deliver the vaccine to health workers up to 10 hours a day, seven days a week.
I am a health worker. How can I access the programme?
The first step to accessing a vaccine is to register on the country’s Electronic Vaccination Data System (EVDS). You can access the system by visiting this website: https://vaccine.enroll.health.gov.za
In general, patient-facing health workers will receive priority access to vaccines.
Non-clinical staff and traditional healers are also eligible to register on the EVDS system.
You will receive an SMS alert with a vaccination voucher and details on what vaccination centre to attend and when.
You will also be asked to read an information sheet and provide advanced consent for vaccination before your visit.
On the day of your vaccination, you will need to arrive with your voucher and an identity document.
Why do I need to sign consent for the vaccine?
Consent to be vaccinated against COVID-19 is being sought in most national vaccination campaigns around the world. This is common when vaccines are provided under Emergency Use Authorisation mechanisms.
You will also be asked to provide consent for the Johnson & Johnson vaccine as part of the early access arrangement through the Sisonke programme.
The information has been loaded on the EVDS system and you will need to read it and agree to receive it before you attend your vaccination visit. Staff will also recheck this with you to attend your vaccination.
It also includes consent to access your medical records to monitor any future episodes of COVID-19 or hospitalisations.
This information will include where appropriate, your ID number, name, date of birth, postcode and contact details.
This information will be kept safe and only Sisonke researchers will monitor the effectiveness of the vaccination programme and access secure and anonymised data.
Are there any reasons why you will not be able to access the programme?
The vaccine is unavailable to pregnant women during this initial stage, which is a common practice when vaccines are not yet fully licensed, such as during clinical trials.
This is done because at this stage there is very little information on the safety of the Johnson & Johnson vaccine in pregnancy.
It is highly expected that the jab will be found to be safe in this population and that pregnant women will become eligible for vaccination in the coming months.
Similar vaccines to the Johnson & Johnson COVID-19 vaccine have shown no safety concerns in pregnancy.
Access to COVID-19 vaccination for pregnant women is likely to be made available in the coming months.
However, the vaccine can be safely used in breastfeeding women.
Department speeds up issuing water use licenses
The Department of Water and Sanitation (DWS) says it is fast tracking water use licence applications to ensure they are finalised within 90 days, in line with President Cyril Ramaphosa’s pronouncement in the State of the Nation Address, last week.
The acting Chief Director of Water Use License Administration in the department, Roxanne Naidoo, on Thursday said they have undertaken preparatory work including re-engineering the current business process to cut it down from 300 days to 90 days.
“This requires a thorough review of the current process and making the necessary changes, in accordance with the relevant sections of the National Water Act.
“The second activity comprises reconfiguring the E-WULAAS (the online application portal) to align it with the revised process,” Naidoo said.
The department, said Naidoo, will immediately commence with workshops in various provinces to capacitate and educate applicants about the new business process. The workshops will be held in February and March.
“Starting from April 2021, all applications that will be submitted and accompanied by all the requisite administrative and technical information will be finalised within the 90 days turnaround time,” Naidoo said.
While noting that human capacity is a challenge, Naidoo said the department is recruiting additional officials in order to fast track the processing of water use licence applications.
Naidoo called on prospective applicants to provide adequate information so that their applications can be issued timeously.
EPWP creates over 500 000 jobs in 2020
The Expanded Public Works Programme (EPWP) has between April 2020 and December 2020 created 515 862 work opportunities for the poor and unemployed South Africans.
According to the Department of Public Works and Infrastructure (DPWI), all four Sectors of the EPWP – Infrastructure, Environment & Culture, Social and Non-State – contributed to these opportunities, with the largest numbers coming from Infrastructure with 208 505 work opportunities, followed by Social with 164 275 work opportunities.
“The EPWP Environment & Culture and Non-State sectors created 108 762 and 34 320, respectively,” the department said on Thursday.
KwaZulu-Natal and the Eastern Cape continue to lead in the creation of EPWP opportunities, the department said, adding that the two provinces created 144 614 and 108 279, respectively.
The performance by other provinces between 1 April 2020 to December 2020 is as follows:
- Eastern Cape 108 279
- Free State 29 343
- Gauteng 50 917
- KwaZulu-Natal 144 614
- Limpopo 62 715
- Mpumalanga 27 386
- North West 31 754
- Northern Cape 13 875
- Western Cape 46 979
Despite challenges posed by COVID-19, the EPWP continues to create the much needed work and training opportunities for the poor and unemployed South Africans.
EPWP acting Deputy Director General Carmen Joy-Abrahams indicated that the EPWP will continue with initiatives to stimulate employment opportunities and improve livelihoods beyond COVID-19.
The department said all spheres of government and State-Owned Entities are expected to implement the Programme.
EPWP beneficiaries
EPWP beneficiaries work in different projects such as Community Work Programme (CWP), Early Childhood Development Programmes, Home Community Based Care Programmes, Extra School Support Programmes, Working on Fire, Working for Water and Roads Maintenance Projects, among others.
Through various skills and training that the participants receive from the EPWP, they stand better chances to enter the formal job market and/or become entrepreneurs.
KZN rolls out COVID-19 vaccination
KwaZulu-Natal Premier Sihle Zikalala has officially unveiled the COVID-19 vaccine rollout programme in the province, which will see frontline healthcare workers get vaccinated with the newly acquired Johnson and Johnson vaccine.
This follows the launch of the country’s vaccination programme by President Cyril Ramaphosa and Health Minister, Dr Zweli Mkhize, in Cape Town, on Wednesday.
A total of 80 000 doses of the Johnson and Johnson vaccine arrived in South Africa on Tuesday evening.
Zikalala, who was accompanied by Health MEC, Nomagugu Simelane-Zulu, received the first batch of the vaccine on Thursday, where healthcare workers were vaccinated at Prince Mshiyeni Memorial Hospital in Umlazi, south of Durban.
Speaking at the provincial vaccine rollout launch, Zikalala said the arrival of the vaccine in the province is an important step towards rebuilding lives.
“This vaccine is eagerly awaited in all parts of the provinces and if it was up to us, we would have administered it to all healthcare workers in the province on the same day so that they are all protected. For now, we have to make do with what we have,” Zikalala said.
The Premier urged healthcare workers to take the vaccine, ensuring them that government would never subject them to a vaccine that would be detrimental to their lives.
Simelane-Zulu said the province has laid the groundwork for the vaccine rollout.
“We have a continuous education programme and we are conducting webinars with all our stakeholders, including our traditional health practitioners.
“We had planned to vaccinate all healthcare workers within four days, but we had to revise the plan according to the amount of vaccines we get. The good thing about Johnson and Johnson vaccine is that it’s a once-off,” Simelane-Zulu said.
Unions welcome vaccine
Mlungisi Ndlovu, from the KwaZulu-Natal Public Servants Association of South Africa (PSA), welcomed the arrival of the vaccine in the province.
“Our people in KZN and healthcare workers need the vaccine. We must speed up the process of vaccinating people before the third wave arrives in order to reduce deaths,” Ndlovu said.
Democratic Nursing Organisation of South Africa (DENOSA) KZN Provincial Secretary, Mandla Shabangu, also welcomed the arrival of the vaccine and urged healthcare workers to vaccinate.
“With the arrival of the vaccine, our members are excited and enjoy coming to work. We want to fast-track how it is rolled out to other members.
“While we support the view that it’s not compulsory to vaccinate, we encourage our members to take the vaccine,” Shabangu said.
First Lady calls for child hunger to be addressed
South Africa’s First Lady, Dr Tshepo Motsepe, has called on the Ministers responsible for food and nutrition security to strengthen food safety nets for children.
Motsepe made the plea on Thursday, when she delivered the opening address at the launch of the 2020 South African Child Gauge, hosted by the University of Cape Town’s Children’s Institute.
The First Lady said there is a need to strengthen the food security nets for children, including the National School Nutrition Programme, the Early Child Development subsidy, campaigns for exclusive breastfeeding, programmes of food fortification and food supplementation as well as social protection policies.
“And we need these to be effectively implemented and adequately resourced,” said Motsepe.
She added that in line with the recommendations of the United Nations and in support of the Department of Health’s Nutrition Policy, prioritising children and safeguarding their food and nutrition security was “imperative”.
This, she said, would ensure the protection of their health, education and wellbeing, more so now given the negative impacts of COVID-19.
The plight of children has remained a passion of Motsepe in her various capacities and years of experience in health and development work.
“Long before the disruption of the COVID-19 pandemic, we as a country, have been acutely aware that a significant number of South Africans do not have access to sufficient food and go hungry on a daily basis.
“The extent of child malnutrition in South Africa has often been documented and it is with distress that we learn of the deteriorating situation, exacerbated by the COVID-19 pandemic and subsequent lockdown restrictions.”
She bemoaned the country’s child stunting rates that show South Africa is lagging behind in nurturing children.
“Stunting reflects chronic under nutrition and a lack of adequate food to promote optimal growth and development,” said Motsepe. “Our national demographic health survey confirmed that the proportion of children who are stunted has not changed since 1993.”
The negative impact that COVID-19 has had on household earnings and on the prices of basic foods paint a very dim picture regarding any progress to addressing chronic malnutrition of children.
“We salute the efforts and initiatives that communities and civil society have embarked on to bring relief to the hungry,” she said, adding that while the efforts of organisations and individuals were commendable, much more needed to be done.
“We cannot turn our eyes away from images that mirror the gravity of hunger in our society. It is saddening to see people standing in long winding queues for food, as we have seen in media reports,” she said.
The National Department of Health Ministerial Committee for the Morbidity and Mortality of Children has found that malnutrition remains a significant underlying cause of child mortality which is linked to one third of all child in-hospital deaths.
Motsepe said while society may be distraught by these hospital-based findings, the larger burden of malnutrition lies in communities and in families, often hidden until later in the life of affected children.
The First Lady said malnutrition casts a long shadow on children and their futures. It robs them of health and wellbeing and condemns them to continued ill-health while undermining their chances to learn, to earn and to escape poverty.
“It is a slow violence against our children, and we cannot thrive as a country when our children are shackled to a life of hunger and malnutrition,” she said.
She warned that hungry children will go in search of food, leading them onto the streets and unsafe environments.
“Children who are hungry cannot concentrate and learn. Children who are hungry become vulnerable to negative elements who will feed and manipulate them,” she said.
Addressing hunger and malnutrition is not just a health issue, hunger and malnutrition have negative social and economic impacts. “Not enough food in the home has a negative influence on family dynamics and not having enough food as a community leads to instability and violence.”
Motshepe urged society to strive to ensure that no child goes to bed hungry.
She congratulated the 2020 Child Gauge Team for its hard work in documenting the plight of children’s food security and nutrition by providing its latest evidence.
First day of vaccine roll-out programme a success
The Health Department has given South Africa the thumbs up as the country began its first round of the vaccination rollout programme on Wednesday.
Yesterday, the country watched as healthcare workers lined-up to receive the shot of the 80 0000 Johnson & Johnson vaccines that landed on Tuesday, alongside President Cyril Ramaphosa and Health Minister, Dr Zweli Mkhize.
“Day one of the first phase of COVID-19 Vaccine Rollout to health workers was a huge success today,” the Health Department said on Wednesday.
The department said South Africa secured the single-dose Johnson & Johnson vaccine for use in the Sisonke ‘Together’ study, which will be distributed to up to 500 000 healthcare workers in 18 public sector hospitals across all nine provinces.
Meanwhile, the organised labour leadership is expected to receive their shots as a demonstration of confidence in the safety and efficacy of the vaccine at Steve Biko Academic Hospital today.
“These are leaders who are health workers themselves,” the department explained.
The State reiterated that protecting frontline workers remains an urgent priority, who are three to four times more likely to contract COVID-19 than the general population.
Since the outbreak, the department has reported 40 000 South African health workers who have contracted COVID-19, while 6 473 have been hospitalised, with 663 deaths.
Oxford-AstraZeneca
South Africa’s rollout of the Oxford-AstraZeneca vaccine was suspended earlier this month after studies released showed that it is less effective against the mutated SARS-CoV-2 501Y.V2 variant, which is common in South Africa.
The department believes that Oxford-AstraZeneca may still play a role in the national programme. However, more research is needed to determine how and when it will be used.
“Vaccination using a safe and efficacious vaccine must start without delay to protect healthcare workers during a likely third wave this winter. The switch to the Johnson & Johnson vaccine allows the country to restart phase one vaccine rollout to keep our health workers safe.”
Safety first
The department said the single dose of the Johnson & Johnson jab protects against severe COVID-19 in South Africa.
“The safety and efficacy of the vaccine were assessed in the international phase three study, ENSEMBLE, which was conducted across Latin America, the United States and South Africa with more than 43 000 participants.”
South Africa had about 7 000 participants in the trial between October 2020 and February 2021, including those who were exposed to the 501Y.V2.
The department explained that the long-term follow-up of study participants is still ongoing, as is required for the process of regulatory approval by the South African Health Products Regulatory Authority (SAHPRA) for commercial or emergency use.
“However, since the safety and efficacy of the vaccine have already been proven, the current rollout is being conducted under The Sisonke Open-Label Program, to assess real-world effectiveness among healthcare workers, who have already been prioritised for vaccination by the South African Government’s COVID-19 Vaccination Strategy.”
The department has once again assured the healthcare workers that the vaccine has been shown to be safe, with no safety concerns reported from past trials or rollouts.
“The ENSEMBLE trial showed excellent protection of a single-dose vaccine against severe COVID-19, including in South Africa, and as such a delay in rolling out the vaccine would be unethical.”
The Sisonke rollout includes collaboration between a team from ENSEMBLE trial, Johnson & Johnson, the National Department of Health and the South African Medical Research Council.
The partners will closely monitor, track and assess the occurrence of hospitalisations and deaths, incidence of severe SARS CoV-2 infections, diversity of breakthrough infections and evaluate vaccine uptake among healthcare workers in South Africa.
“The protection of our healthcare workers through the rollout of the safe and efficacious vaccine is an urgent priority for South Africa.”
SA looks to Asian markets to revive economic growth
International Relations and Cooperation Minister, Dr Naledi Pandor, says government has created a significant footprint in Asian markets as part of efforts to return the country to pre-COVID-19 economic growth.
The Minister said this when she participated in the debate on the State of the Nation Address on Wednesday.
“We will identify new opportunities and expand those that have benefit for South Africa. In pursuit of this objective, South Africa has created a significant footprint in Asia, which is the continent showing the most promise of a speedy return to pre-COVID-19 levels of economic growth.
“Last year, South Africa acceded to the Treaty of Amity and Cooperation to take up significant trade and development opportunities available in this region. We will also benefit from the Regional Comprehensive Economic Partnership (RCEP) of these countries. This partnership created the world’s biggest trading bloc, estimated to account for about US $26 trillion or 30% of global GDP, and 28% of global trade,” Pandor said.
The Minister said in response to the President’s call of intensifying efforts to stimulate growth, government is working tirelessly to raise South Africa’s global visibility by also “promoting [South Africa] as the best place to be, to do business, to visit, to work, to study and to live”.
“We are also responding to new opportunities and harnessing the collective capabilities of DIRCO’s resources both at home and abroad,” Pandor said.
South Africa, the Minister said, will leverage engagement with the Association of Southeast Asian Nations (ASEAN) to enhance mutually beneficial trade, investment and tourism ties, and to support skills development and training for South Africans.
Pandor said South Africa secured significant beef export opportunities to the Malaysian market as of November 2020.
“Market access was also obtained for fruits to Thailand. Product protocols are being negotiated in the region and are at an advanced stage. These include table grapes to Vietnam and South Korea, avocadoes to India and Japan, and pears to China and India.
“Total two-way trade with Asia and the Middle East (inclusive of Oceania) breached the important milestone of R1 trillion, for the first time, in 2019 and is set to grow further.”
Many companies from the Asian region have made significant investment pledges during the President’s investment Summits and State Visits.
Companies such as Toyota, Isuzu, Tata Motors, Mahindra and Motherson Sumi have expanded their investments in the country.
China has pledged to invest $14 billion, and Saudi Arabia and the United Arab Emirates $10 billion each.
“We aim to build on these successes by ensuring growing global trade, which is one of the best ways to fight poverty, inequality and unemployment in our country and our continent,” said Pandor.
